Covid-19 vaccine 90 percent effective in first analysis, Pfizer says
Thứ Hai, 09/11/2020, 23:27
The drug company Pfizer said Monday that early analysis shows its Covid-19 vaccine candidate for Covid-19 is more than 90 percent effective in preventing infection.
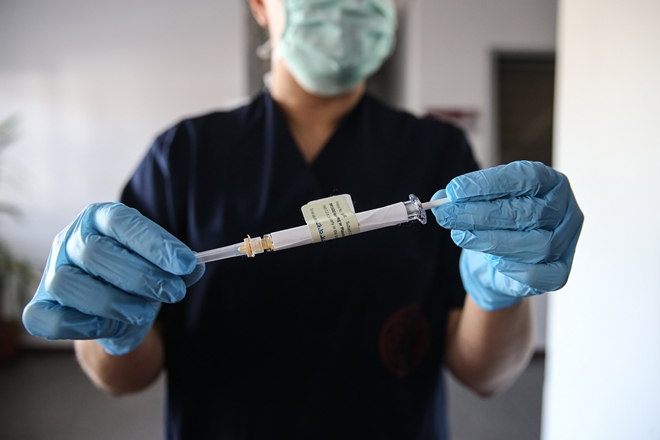 |
| A healthcare worker with a syringe of the vaccine developed by Pfizer and BioNTech at a hospital in Ankara, Turkey on Oct. 27, 2020. Getty Images. |
The drug company Pfizer said Monday that early analysis showed its Covid-19 vaccine candidate for Covid-19 is more than 90 percent effective in preventing infection.
It said no serious safety concerns were observed in the trial, and that it would apply for emergency use authorization to the U.S. Food and Drug Administration after a key safety hurdle was passed, likely in mid-November.
"Today is a great day for science and humanity," Albert Bourla, Pfizer's chairman and chief executive, said in a statement. "We are reaching this critical milestone in our vaccine development program at a time when the world needs it most with infection rates setting new records, hospitals nearing over-capacity and economies struggling to reopen."
NBC

