Local Molnupiravir manufacturers to be licensed soon
Health professionals of the Ministry of Health are due to meet on January 5 to consider licensing local pharmaceutical companies that produce Molnupiravir for COVID-19 treatment.
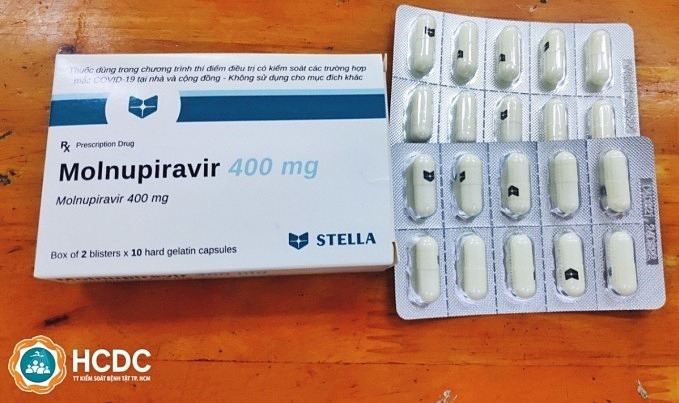
The Advisory Council for the Registration of Circulation of Drugs and Medicinal Ingredients are to consider four out of 10 pharmaceutical companies that have sent requests for the license. The remaining six companies are required to hand in additional documents.
Molnupiravir, the antiviral drug, has been approved for treating COVID-19 patients with mild to moderate symptoms. Studies show the medication can help reduce the numbers of hospitalisations and deaths in COVID-19 patients.
The Ministry of Health has allocated a total of more than 300,000 doses of Molnupiravir to a pilot program for the controlled treatment of mild COVID-19 cases at home. The program was first implemented in Ho Chi Minh City and has now been replicated to 51 pandemic hit localities nationwide.
Preliminary reports show almost 100% of patients taking the drug had a low viral load, a very low rate of severe disease, and no deaths.

